Cascade Pipeline: TARGETING DISEASES DRIVEN BY THE PRESENCE OF HYPOXIA
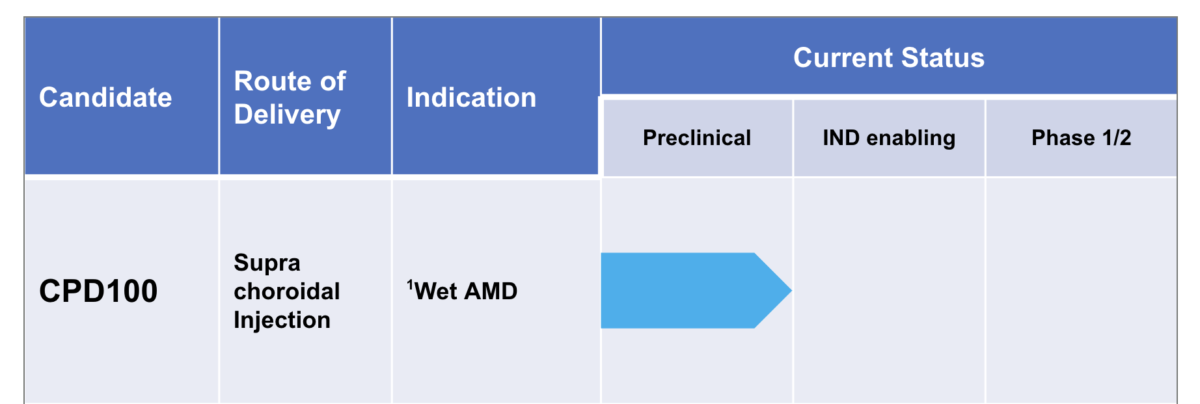
CPD100: Lead Program In Ophthalmology
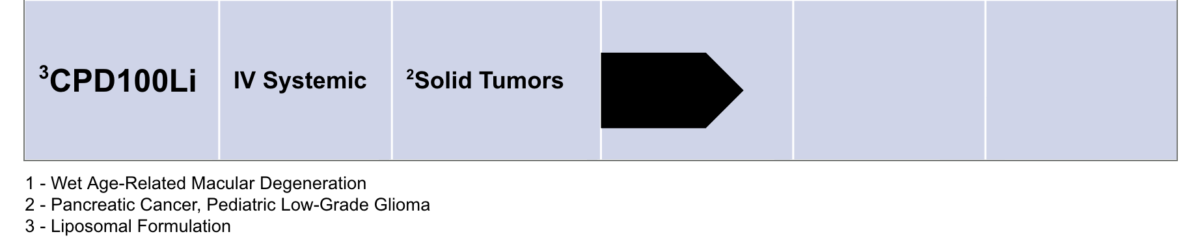
CPD100Li: Program In Oncology
Cascade Development Programs
CPD100 in Wet Age-Related Macular Degeneration (wet AMD)
Executive Summary
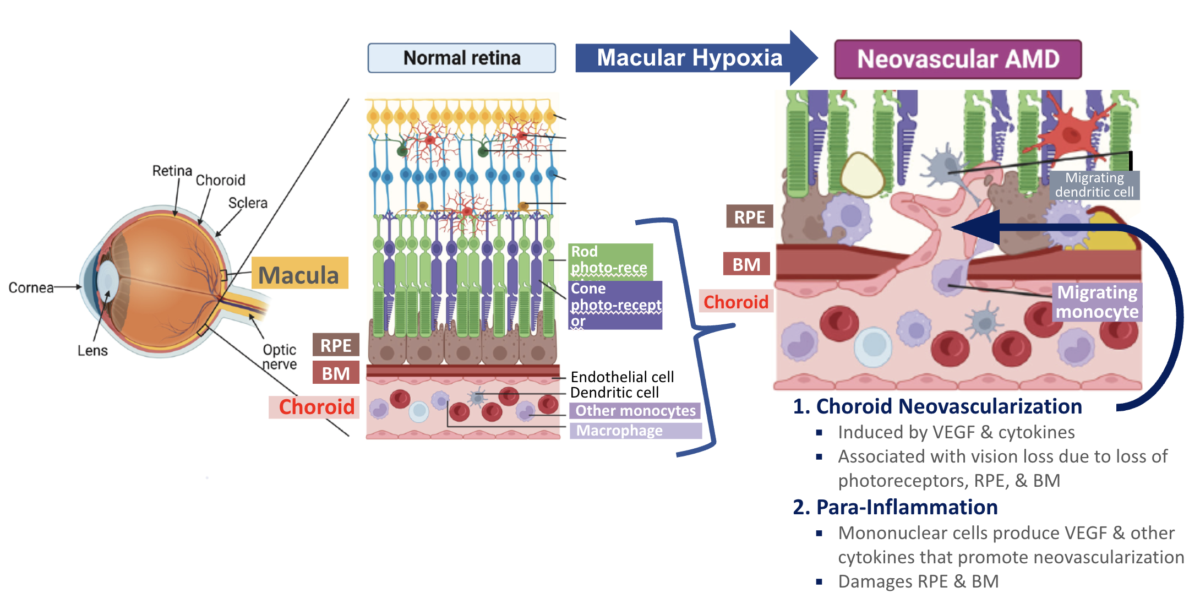
- Wet age‑related macular degeneration (wet AMD) develops primarily from hypoxia-driven vascular endothelial growth factor (VEGF) upregulation and secretion by mononuclear phagocytes, leading to abnormal blood vessel growth (neovascularization) and leakage under the retina, rapidly damaging central vision
- Medicare spends >$4B per year on drugs to treat AMD-related neovascularization
- CPD100 is activated under hypoxia to release vinblastine, which exerts dual anti-angiogenic effects:
- Suppression of abnormal blood vessel growth,¹ and
- Reduction of mononuclear phagocytes and other cells that stimulate excess VEGF production²,³
- Suprachoroidal administration of CPD100 has the potential to become the first targeted therapy to selectively inhibit angiogenesis in wet AMD through a dual mechanism of action
Wet AMD: Evolution in the Ageing Eye
About Wet AMD
- ~2M Americans affected; rapid, permanent central vision loss
- Hypoxia → excess VEGF → choroid neovascularization + mononuclear migration → vascular leakage
- Medicare spends >$4B/year on anti VEGF drugs, standard of care
- Current therapies are palliative, not disease modifying
- Multiple blockbuster intravitreal-anti VEGF drugs (Eylea®, Lucentis®, Vabysmo®)
- Strong investor and pharma activity (e.g., EyeBio/Merck $1.3B upfront acquistion)
Limitations of Anti VEGF Therapies
- Require intravitreal injections with limited targeting to the retina and choroid (painful)
- Monthly loading + q2–4 month maintenance
- Do not address hypoxia, direct action on growth of endothelial cells or on VEGF‑producing immune cells
- Risks: retinal tear, hemorrhage, cataract, uveitis, IOP elevation
CPD100 MECHANISM OF ACTION
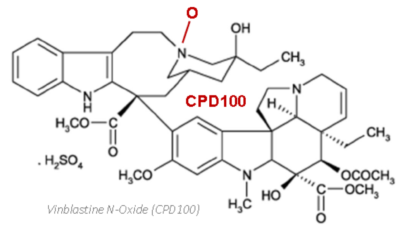
- Vinblastine N‑oxide is a prodrug converted to vinblastine (VBL) only in hypoxic tissue
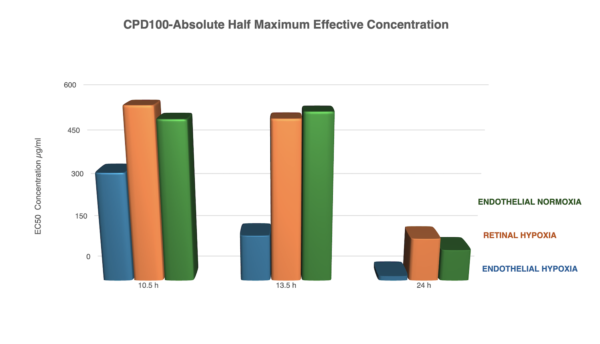
- Endothelial cells – but not retinal cell -are highly sensitive to low doses of CPD100 under hypoxia → rapid shutdown of abnormal vessel growth and spare toxicity to retina cells
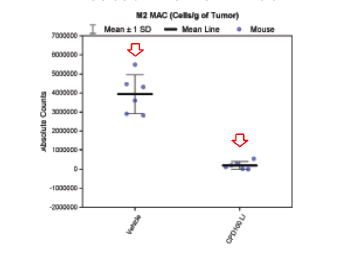
- Mononuclear phagocytes (M2) reduced → upstream VEGF suppression²,³
- Potential for more durable and more complete disease control
GROWTH INHIBITION OF ENDOTHELIAL CELLS EXPOSED TO CPD100
REDUCTION OF M2 CELLS IN PRECLINICAL MODELS TREATED WITH CPD100
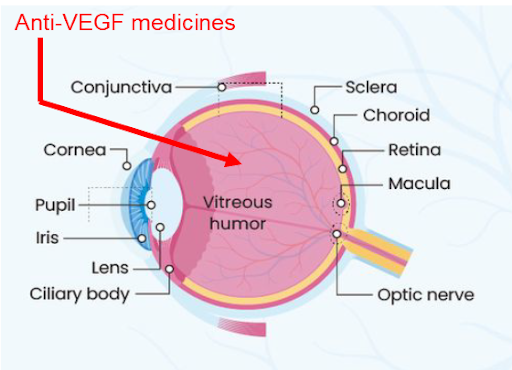
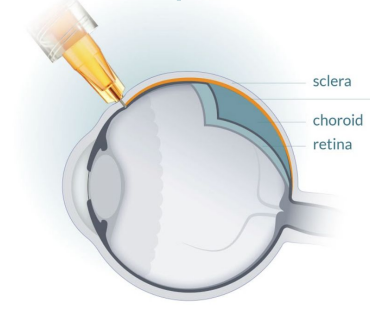
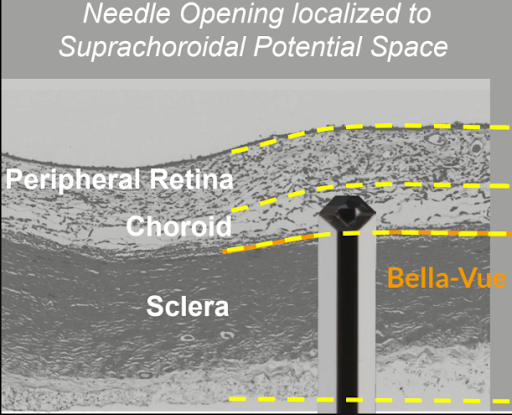
CPD100 SUPRACHOROIDAL ADMINISTRATION FOR WET AMD
- Direct access to choroid, the site of pathology
- Minimizes vitreous exposure and systemic toxicity
- FDA‑cleared micro‑injection technology (e.g., Bella-Vue / Uneedle) validated in AMD trials
- Enables safer and more targeted therapy to the choroid
- Growing clinical adoption of suprachoroidal injection
- Easy and reliable to use — 100% user satisfaction; single microneedle length¹⁰
Commercial Opportunity for CPD100 in Wet AMD
- Wet AMD prevalence: ~2M US patients
- CPD100 advantages:
- Only drug with dual MOA: anti-neovascularization + anti-inflammatory
- Potential to improve vision & decrease frequency of administration
- Suprachoroidal administration avoids intravitreal morbidity
- Potential ≥20% market share in mid‑2030s
- Expansion into other indications:
- Predominant Antiangiogenic Effect
- Diabetic macular edema (DME)
- Macular edema following retinal vein occlusion (MEfRVO)
- Predominant Anti-Inflammatory Effect
- Dry AMD – Geographic atrophy
- Prior retinal transplantation in retinitis pigmentosa, AMD, etc.
- Oncology
- Retinoblastoma
- Uveal melanoma (+ Ipilimumab)
CPD100Li in Hypoxia-Prone Tumors
Hypoxic regions within solid tumors drive profound biological instability, including impaired genome integrity, increased chromosomal instability, elevated mutagenesis, reduced immune surveillance, and heterogeneous perfusion across the tumor microenvironment. Collectively, these pressures promote the emergence of invasive, metastatic clones and confer resistance to local therapies (surgery, radiotherapy) as well as systemic treatments (chemotherapy, targeted agents, and immunotherapies). Targeting these hypoxia‑induced changes is therefore essential to interrupt tumor evolution and reduce the lethality associated with metastatic disease.
CPD100Li is a liposomal formulation of CPD100 designed for systemic administration to counteract the consequences of a hypoxic tumor microenvironment and selectively eliminate hypoxia‑driven malignant clones, reduce tumor-driven angiogenesis and increase immune surveillance.
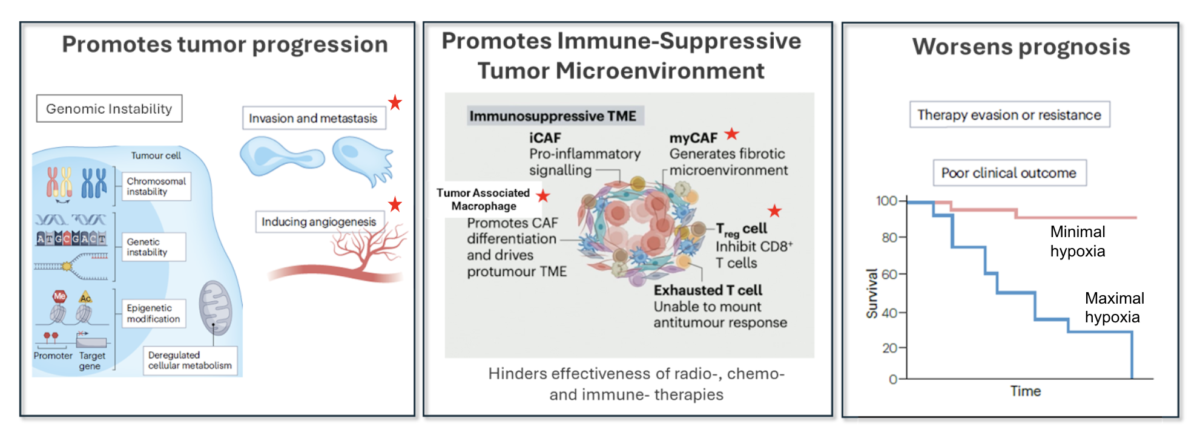
Supports the foundation of our development of CPD100Li
Hypoxia is a common feature of most solid tumors
| Tumor Type | Normal Tissue Median Partial Oxygen Pressure in mm of Mercury |
Tumor Tissue Median Partial Oxygen Pressure in mm of Mercury (# of patients) |
|---|---|---|
| Pancreas | 57 | 2 (8 pts) |
| Brain | 26 | 13 (104 pts) |
| Lung | N/A | 16 (26 pts) |
| Breast | 52 | 10 (212 pts) |
| Cervix | 42 | 9 (730 pts) |
Oxygen partial pressure (pO2) measured in mm of mercury (mm Hg) using electrode probes
CPD100Li MECHANISM OF ACTION IN HYPOXIA-PRONE TUMORS
CPD100Li is a liposomal formulation of vinblastine N‑oxide designed for intravenous administration. The liposomal encapsulation extends the circulating half‑life of the prodrug compared with vinblastine or non‑liposomal CPD100, enabling greater tumor exposure, less frequent dosing, and an improved systemic safety profile.
Once within hypoxic tumor regions, CPD100Li undergoes preferential activation to regenerate vinblastine. Vinblastine is a well‑characterized inhibitor of microtubule assembly that induces cell‑cycle arrest and apoptosis in proliferating tumor cells.
Beyond its cytotoxic activity, vinblastine also modulates the tumor microenvironment by reducing immunosuppressive M2 macrophages. This shift toward a less suppressive microenvironment may enhance responsiveness to immunotherapies, checkpoint inhibitors, targeted agents, and cellular therapies.
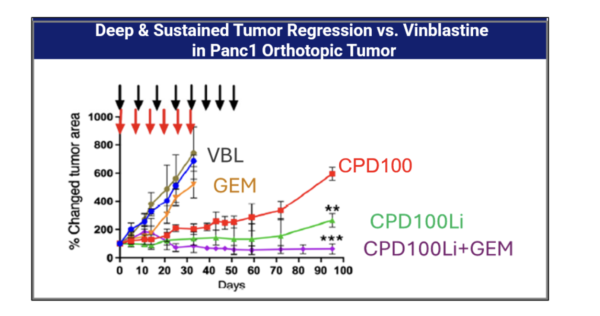
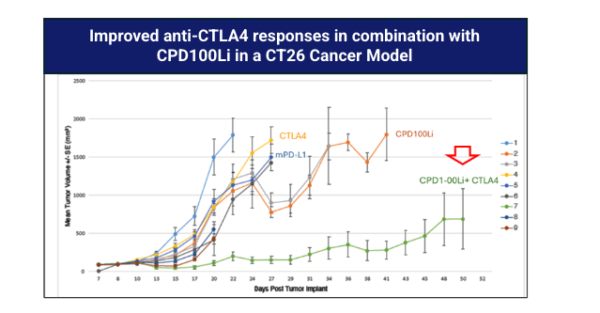
CPD100Li in-vivo activity as monoTx and in combination with Gemcitabine and anti-CTLA4
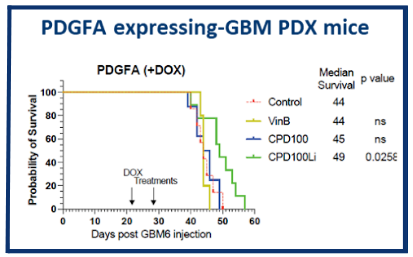
In pancreatic cancer (Panc1) models, CPD100Li shows stronger antitumor activity than vinblastine alone and demonstrates marked synergy with gemcitabine. Similar effects are seen in the CT26 colorectal cancer model, where CPD100Li enhances responses both as a monotherapy and in combination with anti‑CTLA‑4 therapy. CPD100Li has also extended survival in a glioma model. Several tumor types are known to be hypoxic, aligning directly with CPD100Li’s hypoxia‑activated mechanism and its potential used alongside with chemotherapy and immunotherapies.
CPD100Li in Glioma Cancer Model